  
Historically, krypton was widely considered to be chemically inert however, since the 1960’s, studies have found several compounds of krypton – including krypton diflouride and an oxyacid salt containing krypton. Krypton Electron configuration Lewis structure Atom, american element, chemical Element, white png PNG keywords PNG info resize png Relevant png images. Krypton is most often combined with other rare gases to make fluorescent lamps. It consists of 8 electrons in its valence orbit. The element’s gas concentration in earth’s atmosphere is about 1 ppm and is isolated by fractioning liquid air. Krypton Atomic Number: 36, Atomic Radius: Atomic Symbol: Kr, Melting Point: Atomic Weight: 83.79, Boiling Point: Electron Configuration: Ar4s23d104p. Compounds and Isotopes The Electron Configuration of Krypton is 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d10, 4p6 Krypton, a noble gas, has 8 Valence electrons already. Krypton: Having atomic number 36, krypton is used in lighting and high speed photography. 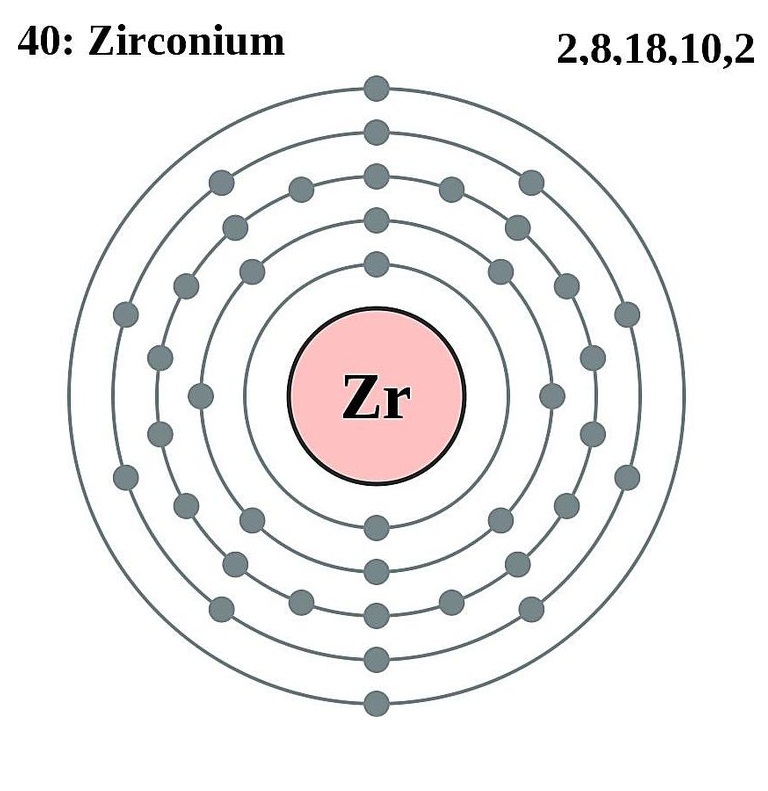
Start your 48-hour free trial to unlock this answer and. This electron configuration shows that the zirconium ion has acquired electron configuration of krypton and it achieves an octave full stable electron configuration. This totals up to 36 electrons, which is the atomic number of krypton. The vibrant green and orange spectral-signatured element is considered a noble gas due to its low chemical reactivity. The electron configuration shows that the zirconium ion (Zr 4+) has four shells and the last shell has eight electrons. The element was found in residue of the evaporated components of liquid air by W. Krypton, named after the Greek word kryptos, meaning “hidden”, was discovered in 1898. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
